In the early 1990s, the founder and chairman of our headquarters at InBody Co., Ltd. in Seoul, South Korea, Dr. Kichul Cha, recognized that the available BIA devices were limited and faulty. They were often inaccurate, and from a medical standpoint, the data sets were heavily reliant on empirical estimations that may not represent all patients. This approach might lead to inaccurate representations of patient health status and be less sensitive to treatment progress over time. Drawing from his background in mechanical engineering, he set to work to design something better.
In 1996, he founded InBody. Two years later, the first InBody device was born. InBody’s 4 core technologies set us apart from other BIA devices. InBody has direct segmental measurement that separately measures the trunk, arms, and legs, which is important for accurate body composition. The use of multiple frequencies measures total body water, which includes fluid both inside and outside of body cells. Our 8-point tactile electrode system provides 99% reproducibility across tests. InBody does not use empirical estimations in measured values, and we are correlated at 98% with DEXA. InBody was also validated in diverse populations.
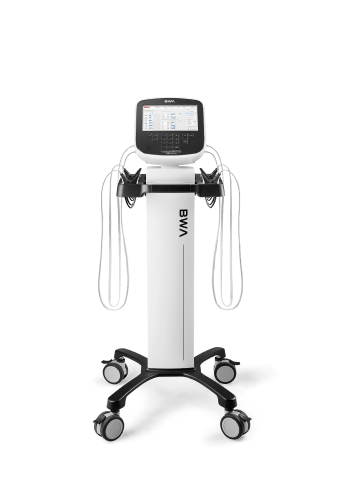
Today, InBody has grown from a small biotech startup in South Korea to a multinational corporation with branches and distributors in more than 80 countries. Because of the growing demand for our devices in the critical ill and the over 6000 peer-reviewed publications involving InBody, InBody BWA Inc was born.
BWA’s sole mission is to improve lives through improved standard of care for the critically ill. Guided by our body water analysis, clinicians and researchers will be bringing more precision to patient care.
Our job now is to simply share – InBody BWA everywhere.